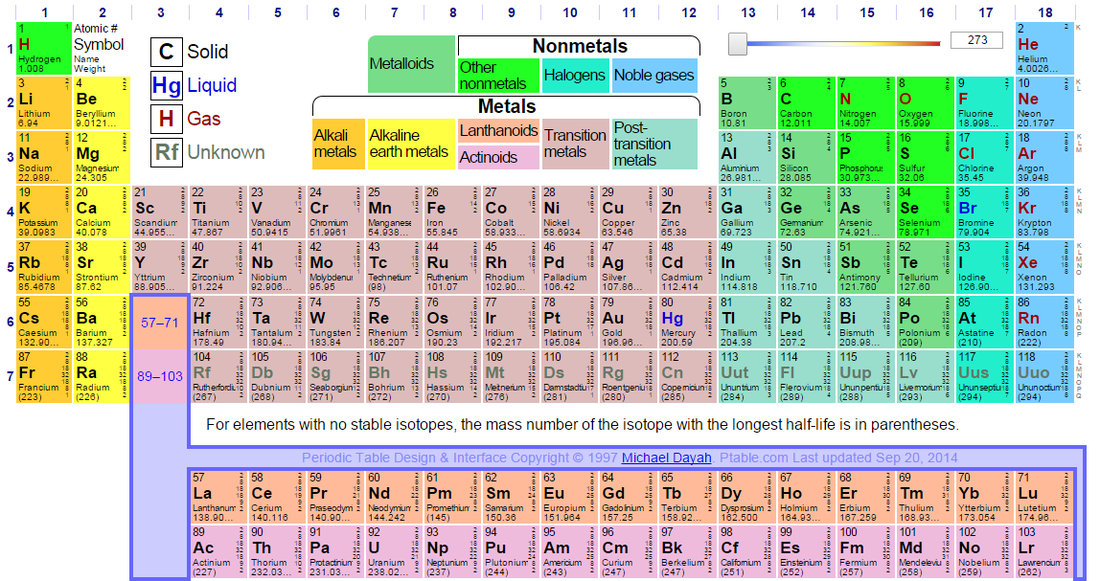
The periodic table
The periodic table is possibly one of the most important tools in chemistry available. It lists every single known element. These elements are placed in order of their atomic numbers, which is the same as the number of protons or electrons in an atom. The elements are arranged in groups and periods. Groups are the columns of elements down the table, periods are the rows. The periodic table can be used by chemists to observe patterns and relationships between the elements. It also displays what series an element is from, listed below:
· Alkali metals
· Alkaline earth metals
· Lanthanoids
· Actinoids
· Transition metals
· Post-transition metals
· Metalloids
· Noble gases
· Halogens
· Other non-metals
· Alkali metals
· Alkaline earth metals
· Lanthanoids
· Actinoids
· Transition metals
· Post-transition metals
· Metalloids
· Noble gases
· Halogens
· Other non-metals
Boron
Boron, number 5 on the periodic table, is a metalloid, meaning it has properties of a metal and a non-metal. It belongs is period 2 and group 13. Boron was discovered by Sir Humphry Davy, an English chemist, in 1808. Two types of boron can be found found. Amorphous boron is a brown powder and metallic boron is black and hard. Pure boron is used as a dopant (a substance added to semiconductors to change how it behaves with electricity) in the semiconductor industry. Chemical compounds of boron are important as to make strong materials that do not weigh very much, as nontoxic insecticides and preservatives. Plants need boron in them to live. Very small amounts of boron are needed in animal's bodies so that they are healthy. Boron's melting point is 2076'C and the boiling point is 3927'C.
Carbon vs. boron
Boron is rather similar to carbon in many ways. Being right next to each other on the periodic table, they are in the same group, and carbon has only one extra electron than boron. Forms of carbon (primarily) and boron (boron carbide) are the hardest materials on earth, and they are both solids. They each include the same amount of neutrons in their atomic structure – 6 – and are capable of forming covalent bonds. This is due to their valence electrons, but this is where is begins to differ. Boron has 3 valence electrons, carbon has 4. Carbon occurs naturally, whereas boron does not. Finally, boron is classified as a ‘semimetal/metalloid’, carbon is known as an ‘other non-metal’. Having few differences, it is concluded that boron and carbon are very similar.