Bonding
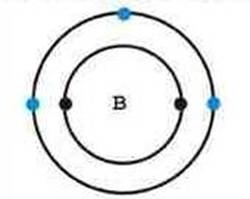 Boron Atom
5+=5-
Boron Atom
5+=5-
An average boron atom contains 5 protons and 5 electrons. The electrons are arranged in shells, with 2 on the first shell and 3 on the second shell.
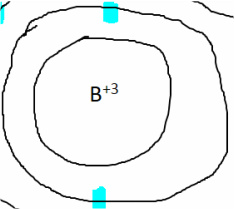 Boron Ion
5+=2-
Boron Ion
5+=2-
To be able to form bonds, a boron atom must first become stable. It has to lose 3 electrons, as its outer (valence) shell is not full, therefore unstable. By losing 3 electrons, boron becomes boron+3.
Types of chemical bonding
Ionic Bonding
In ionic bonding electrons are completely transferred from one atom to another. In the process of losing or gaining electrons, the atoms for ions (charged particles). Positive and negative ions attract one another and bind together to form a new substance. This is called ionic bonding.
Covalent bonding
Covalent bonds are formed between non-metal elements. They bond by sharing valence electrons (electrons on the outer shell). Covalent compounds do not conduct electricity, are usually gases or liquids, and have low melting and boiling points.
Metallic bonding
Metals have loose electrons in the outer shells which form a 'sea' of negative charge around the close-packed positive ions. There are strong electrostatic forces holding these particles together.
In ionic bonding electrons are completely transferred from one atom to another. In the process of losing or gaining electrons, the atoms for ions (charged particles). Positive and negative ions attract one another and bind together to form a new substance. This is called ionic bonding.
Covalent bonding
Covalent bonds are formed between non-metal elements. They bond by sharing valence electrons (electrons on the outer shell). Covalent compounds do not conduct electricity, are usually gases or liquids, and have low melting and boiling points.
Metallic bonding
Metals have loose electrons in the outer shells which form a 'sea' of negative charge around the close-packed positive ions. There are strong electrostatic forces holding these particles together.
Boron and Bonding

Boron is capable of forming ionic and covalent bonds, but cannot form metallic bonds.
What do these boron compounds look like?
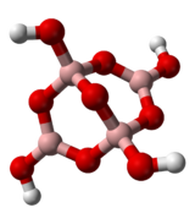 Borax
Borax
An example of a ionic boron bond is sodium borate, commonly known as borax. It is written as [B4O5(OH)4]2−. A borax ion has displayed to the right. Powdered borax is white and can easily be dissolved in water. The melting point is 743'C and the boiling point is 1575'C.
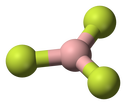 Boron trifluoride
Boron trifluoride
An good example of a covalent boron bond is boron trifluoride, written as BF3.
