History of Atomic theory
Currently, the understanding of atoms and their properties is quite advanced. The quantum mechanical model of an atom has been agreed upon. Atoms are comprised of electrons (negative charge), protons (positive charge) and neutrons (negative charge). The protons and neutrons reside in the nucleus, with the electrons floating around in surrounding shells.
 Democritus, 460 BC
Democritus, 460 BC
Democritus, way back in 460 BC, developed the first idea of atoms and atomic theory. If you break a piece of matter in half, then break it in half again, how many breaks will you have to make before you can break no further? By asking this question, Democritus established that it must end at some point, at the smallest piece of matter. He called these basic matter particles, atoms. Many people dismissed these ideas, including Aristotle, and for over 2000 years nobody did anything further to pursue the subject.
It wasn’t until the 1800’s before atomic theory really began developing. An English chemist, John Dalton performed experiments that proved Democritus’s theory. He speculated that all atoms of one element are identical, but are different from atoms of other elements. Although he did not understand how these atoms were structured, he know that the evidence pointed to something revolutionary and important.
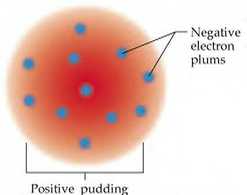
In 1904, another English physicist by the name of JJ Thomson discovered the electron, a particle that makes up an atom. He believed that since electrons had a negative charge, the surrounding matter would be positively charged. Thomson created a model of his theory, and it has been nicknames the ‘Raisin in the Pudding’ model of an atom.
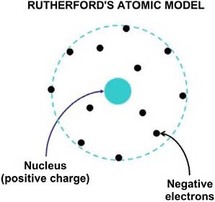 Rutherford's Atomic Model
Rutherford's Atomic Model
Later, in 1911, Ernest Rutherford proposed a new model of an atom. It featured a positively charged nucleus holding most of the atoms' weight, a large space around the nucleus in which the electrons moved, and the nucleus containing positively charged protons. Rutherford also discovered that the number of protons was equal to the number of electrons in an atom. He discovered all this by experimenting with gold foil and alpha particles. Most particles passed through the gold foil suggesting that an atom is largely empty space. Some particles were deflected significantly, suggesting that the positive charge of an atom must be concentrated in a very small sphere.
 Niels Bohr
Niels Bohr
Shortly after, in 1913, Niels Bohr proposed a 'planetary' atom model, meaning that the electrons moved around a fixed nucleus. James Chadwick, in 1932, developed a experiment which included bombarding beryllium with particles, and ended up discovering that Rutherford had failed to observe the neutral particles (neutrons) in the nucleus of an atom. The mass of these neutrons are similar to that of a proton.
